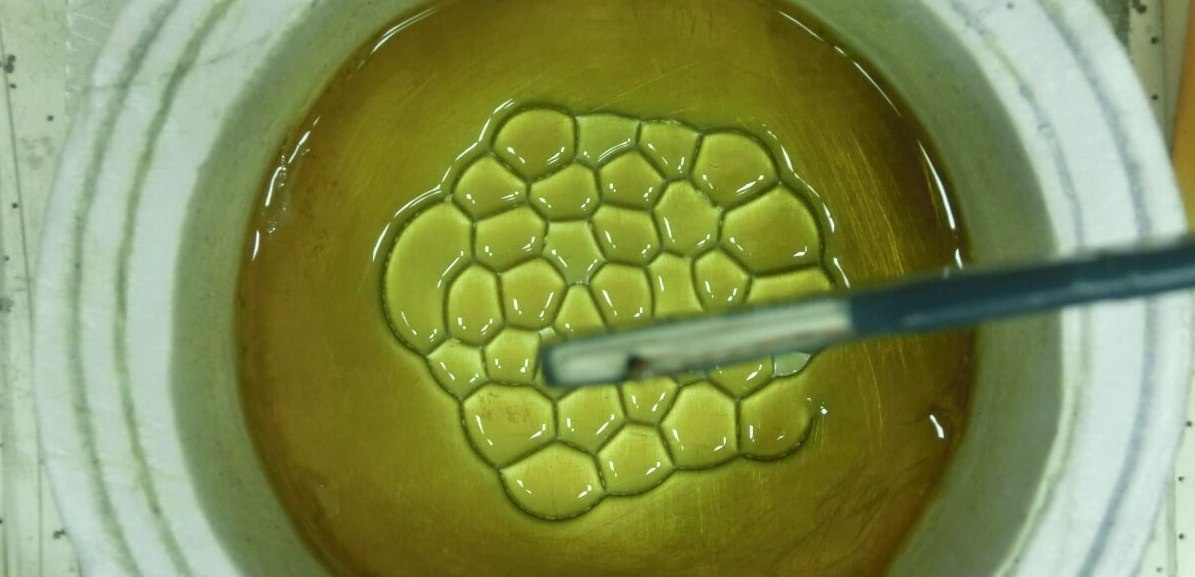
Pour some oil onto a horizontal metal plate and place a vertical metal needle above it. When applied between the needle and the high voltage plate, a cellular structure appears on the surface of the oil.
Let's see why this happens?

The fact is that when we apply a high voltage to the needle, a corona discharge forms on it, which ionizes the air.
Corona discharge photography in the dark
photography in the dark aging photo
aging photo 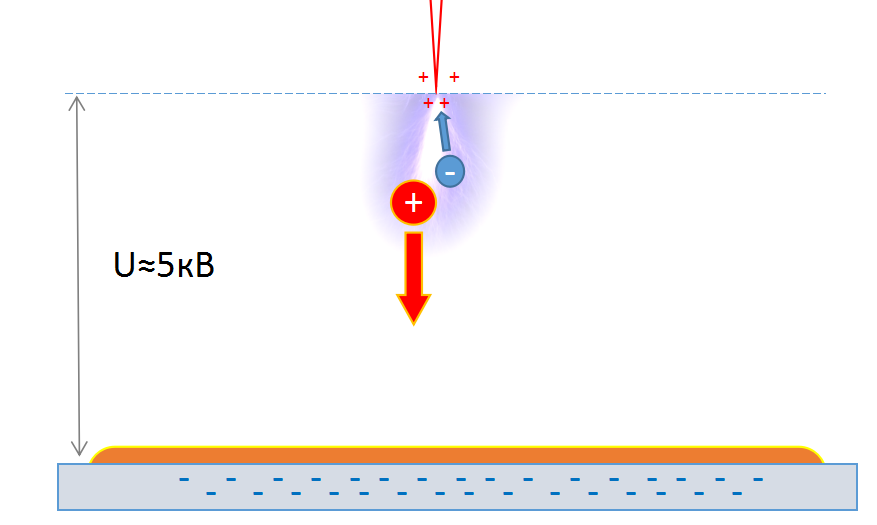
After ionization, we get a positive ion and a negative electron. Since the particles obtained are charged and are in an electric field, a certain force acts on them. If the needle is the anode and the plate is the cathode, then the positive ions will be attracted to the negatively charged plate.
Ion wind
The flame of a match is a plasma, that is, a highly ionized gas.
You can notice the influence of the electric field on the flame.
It can be assumed that the appearance of honeycombs is associated with the action of ions on the oil. This is proved by the following experiment.
But oil is a dielectric, that is, it does not allow ions to pass to the plate. It turns out that the ions remain on the surface of the oil and evenly press on it.
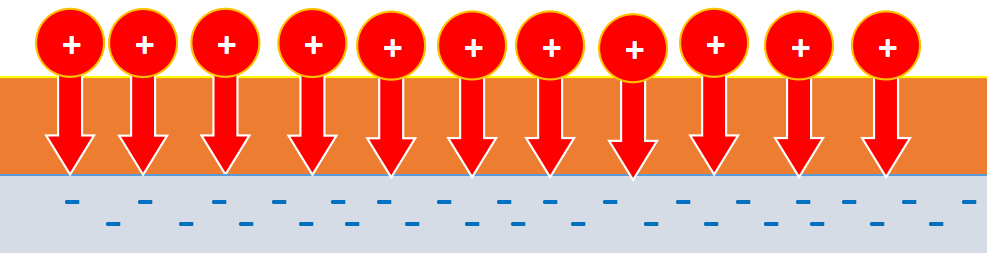
But still, why do we get honeycombs? If we apply a smaller voltage, then instead of cells we will get small pits.
Due to fluctuations, some ions are closer to the plate, hence their force of attraction to the plate increases. They begin to push oil, entraining its molecules. Thereby, causing similar deviations in other places.
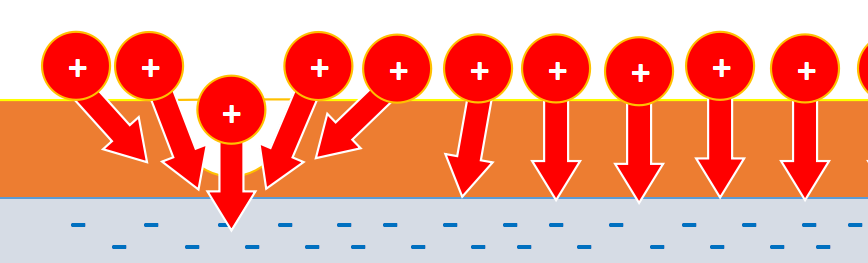
With further increase in voltage, the number of pits and their size increase. And it is energetically advantageous to connect these pits with channels.

What we get in practice.

And what will happen if we pump out the air. It is logical to assume that there will be no honeycomb, since there is nothing to ionize. But no, when pumping, we just cut the air, hence the ion has a longer free path, and it develops a greater speed.
 By reducing the pressure, you can achieve the effect of cells at lower voltages.
By reducing the pressure, you can achieve the effect of cells at lower voltages.At the end of this article, I want to show this phenomenon again.