 Cross-linking
Cross-linking is damage arising in the process of metabolism, and therefore is a normal effect of the normal operation of cellular biochemistry. Different types of glycated molecules, known as
glycation end products (AGE) , accumulate between cells and interact with the
extracellular matrix , linking its various structures. The organization and composition of these structures endows tissues with characteristic properties such as elasticity (skin and blood vessels), strength (cartilage and bone tissue), etc. Cross-links that accumulate in large quantities violate these properties. The mechanisms of violation are different. For example, crosslinks interfere with the free passage of long parallel molecular structures across each other. There is also evidence that glycation products lead to an increase in chronic inflammation, altering cellular function through an
appropriate receptor (RAGE) . Inflammatory processes contribute to the pathology of all the most common age-related diseases.
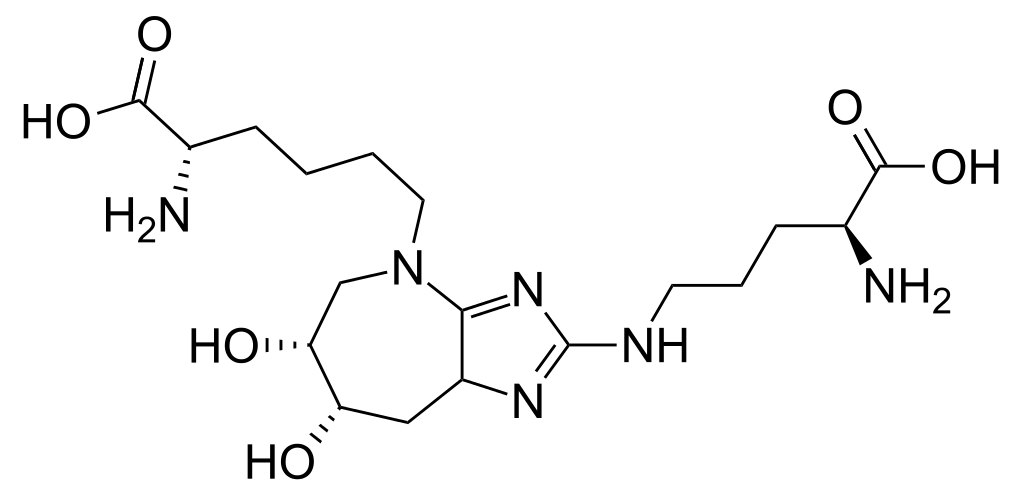
Most of the resulting cross-links are rapidly destroyed, and their role is probably noticeable only in case of metabolic disturbances, for example, in obesity or type 2 diabetes. At the moment, it is believed that the main problem leading to an age-related deterioration of skin elasticity and vascular stiffness is associated with one specific type of cross-linking formed by one type of AGE, the
glucoseepane molecule (glucosepane). Studies show that glucosepan forms the vast majority of cross-links in the body of an elderly person - so strong that the body simply does not have natural mechanisms for their destruction.
Since we are talking only about one molecule, all that is needed to significantly reduce its contribution to aging is one sufficiently effective means capable of destroying this compound. The target drug market is more than half the human population (almost all, over 30 years old). However, a wide community of researchers does not show interest in this task, which is due to the lack of tools for working with glucosean. Any research team that tackles this problem will have to start from scratch, which means that almost everyone who took the time to think about it, eventually chose others that are more accessible to solve the problem. This situation requires philanthropy to get things moving. The only significant source of funding for glucosepane research over the past few years has been the
SENS Research Foundation, thanks to such philanthropists as
Jason Hope , as well as the
support of our community .
However, since the problem described above is too special, in my opinion, it is more likely that the first financial support will be received by another SENS-technology, known as senescent cell clearance. This method is currently
being developed at Oisin Biotechnology , and after it there is a competition for SENS funding between the glucosepane destruction project and
transthyretin amyloid removal therapy with the
mitochondrial DNA restoration project that started a couple of years ago. However, my understanding of the latest trends is a bit behind modernity, so recently I talked with several people working in this field.
Aubrey de Gray (SENS),
David Spiegel (Yale) and
William Baines , who collaborate with a huge number of researchers in various fields, including those in discussion, became my interlocutors. Below is a sketch of the result of these conversations.
Getting glucoseepane is a big step forward.
The laboratory of David Spiegel developed in 2015 a
reliable way to obtain glucoseepane . This is an important event, since people who previously had no opportunity to participate in this field of research can now start their own research projects. It also guarantees, at least in the near future, uniformity in what is meant by glucosepan and its molecular structure.
No complete consensus on glucosean has yet been reached.
The fact that it is glucosepan that is responsible for most of the dangerous crosslinks is increasingly being questioned. However, according to current data, the fight against glucosean as the main goal is justified. David Spiegel is optimistic about this and believes that the right tool will inevitably be found. Aubrey de Gray, however, is more cautious in his assessments, while William Baines is completely disappointed with the poor quality of past research. Of course, after the victory over glucosepan, doubts will be resolved in one direction or another, but for now this is a question of the future. The SENS Foundation is now sponsoring joint research by Jonathan Clark at the
Babraham Institute , aimed at more precise clarification of the role of glucosepan, confirmation of its current ideas about its structure (it’s important to know not only the composition, but also the spatial organization of the molecule, which determines its properties) and the search for other possible molecules responsible for the formation of stable cross-links leading to pathologies.
Drug candidate not found yet
At the moment there is not even an approximate idea of how to find the necessary drug among a huge catalog of existing and developed drugs. The lack of ideas stems from the lack of research on the organization of similar molecules in pharmacology. Selection of a suitable drug is the main obstacle to the creation of a company that draws a line in the fight against glucosean. The job of testing a drug is much easier than finding the right candidate. To analyze its effectiveness, it is sufficient to organize testing of glucosepan levels before and after treatment. One of the standard approaches here is to use
antibodies introduced into the
immune system and interacting with glucosean as markers.
Most likely a suitable candidate will appear in the process of studying bacteria.
Spiegel Lab uses the same approach as used in
the LysoSENS research program . Its essence lies in the fact that among the bacteria are looking for enzymes that can effectively destroy glucosean. These enzymes certainly exist if only because our cemeteries can hardly be called a warehouse of sticky sugar. The fact that LysoSENS’s research in this area was conducted ten years before they found the first applicants for the desired enzyme and received financial support from
Human Rejuvenation Technologies may seem discouraging. However, only in the last few years significant progress has been made in the cultivation of bacteria, and thus, researchers now have an advantage that is not available in the earlier LysoSENS project. One of the open problems so far has been the fact that 99% of all types of bacteria could not be grown in the laboratory. However, after a relatively simple
technological breakthrough, the situation has changed. Everything that has been achieved so far by scientists has been obtained as a result of studying only 1% of bacterial species, but now they can all be subjected to regular research, thus the space for work has increased a hundred times.
Employees of Spiegel's laboratory have already identified and learned how to grow certain types of bacteria that can probably absorb glucoseepan. David Spiegel believes that with the current level of funding, it will take about two years to figure out the mechanism of absorption. It is possible that the presence of a simple enzyme is sufficient for absorption, perhaps the process is more complicated. If it is a single enzyme, then it can quickly become the desired drug, if not, then you will have to research a larger number of bacteria, among which there is a more suitable candidate. This work can progress faster if funding is increased, since research on different bacteria can be conducted in parallel by a large number of researchers. However, the search for funding in this area is a problem in itself.
I note that I omit the difficulties that will arise when extracting the necessary enzymes from bacteria and the production of drugs on their basis. Undesirable effects, such as an adverse reaction of the immune system, are quite likely. This problem cannot be solved by simply placing the necessary preparations in a protective capsule, since its contents are supposed to enter the extracellular space. The list goes on. But all these obstacles seem surmountable, additional work will require the application of new technologies and approaches that may come from other areas of science.
Two models for future commercialization
Starting from this point, there are two possible ways of commercial development. The first is to find a willing risk investor who will invest $ 2 million and wait patiently for two years until David Spiegel, William Baines and Jonathan Clark finish their job. This scenario is implemented in many areas of research, but it requires good connections and a thick wallet. That is why this sort of contract is usually concluded with pharmaceutical companies, as, for example, happened with the development of
transthyretin amyloid clearance therapy based on CPHPCThe second way is to encourage researchers and support them, as we can, with the help of our donations, perhaps for several years, until a suitable enzyme is found. When it comes close to testing the drug in rats and mice, you can organize a startup based on seed funding. This scenario is much simpler in our community — if Oisin Biotechnology was able to get funding from SENS, the anti-glucosean therapy development company could do the same in a couple of years.
Translated by
El Mekhtiyev , SENS Volunteers Group